
Extract the most accurate data from standardized images to provide your product efficacy study with a competitive and scientific edge.
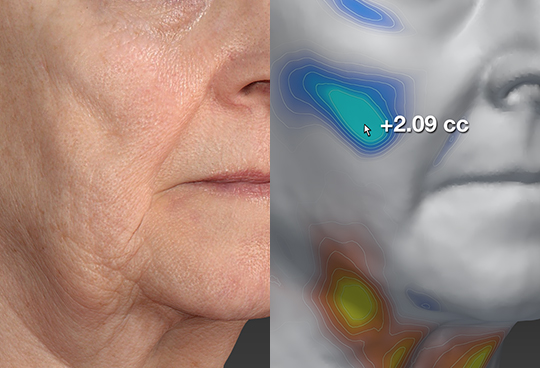
Quantitative Analysis
Objective measurements can provide quantitative data along with the photographic documentation. When used in conjunction with our 2D and 3D standardized photographic equipment, Canfield delivers data such as surface area, volume, skin movement and much more. This data is routinely used for new drug applications, claims substantiations, publications, promotions, and marketing. Imaging data produced in our 21 CFR Part 11 compliant environment is routinely submitted to the FDA and regulatory authorities worldwide in support of both new drug applications and advisory panel meetings.
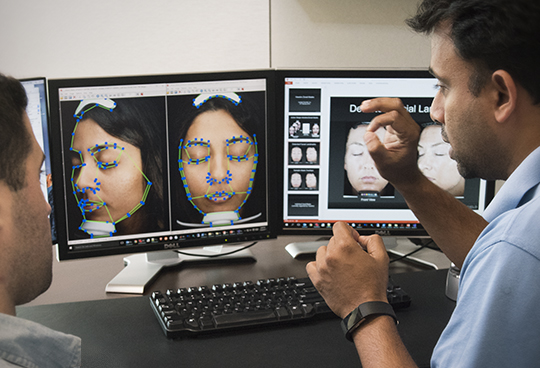
Customized AI
Methods and algorithms are developed and executed by experienced, knowledgeable, scientific imaging PhDs. Work directly with our certified image analysis technicians for a customized analysis methodology specific to your study.



 Steven Kempers, MD,
Steven Kempers, MD, Robert Weiss, MD,
Robert Weiss, MD, Jason D. Bloom, MD,
Jason D. Bloom, MD,